In 2004 Brenda’s son, John Paul, experienced trauma to his front anterior teeth. A strong aesthetic restoration for paediatric dentistry did not exist, which is what sparked the innovation of the 1st prefabricated zirconia crown for primary teeth. Brenda, along with her education in business and experience in the dental industry worked with Dr. Jeff Fisher and many others to bring Sprig to where it is today. After many years of working with Sprig’s founders, the transition to CEO was a natural fit. Once elected in 2017, the first major undertaking was the rebrand. Dental Tribune MEA had the pleasure to interview Brenda Hansen, CEO of Sprig Oral Health Technologies and ask about the future plans.
How does Sprig Oral Health Technologies differentiate itself from other dental manufacturers? What is the core business and the company culture? Who are your customers? Which markets do you cater to?
It starts with our mission. Sprig’s mission is to bring the innovation today’s patients and professionals demand to paediatric dentistry to provide safer, healthier options for the next generation.
Our corporate culture is one of honesty, integrity, respect, continuous improvement and gratitude. These values are what we live by on a day-to-day basis in everything we do. We also consider ourselves to be a pretty fun group. We have been told, that you can feel that type of energy at our events and when interacting with our team. This makes me very proud and I work hard to protect this part of Sprig.
We consider anyone who comes in contact with Sprig to be our customers and this includes our employees. Most of our direct contact is with doctors and their team members. Knowing that we are helping them improve their practice of dentistry is also a driving force for the Sprig Team.
We ultimately serve children and specifically their dental health, knowing that this improves their quality of life. What could be a better purpose than that?
Which is the current, most valuable solution, Sprig provides to paediatric dentists?
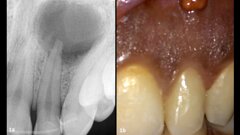
The most valuable solution that Sprig offers is our flagship product, EZCrowns, which is the alternative for an SSC or strip crown. SSC’s and strip crowns have major disadvantages and EZCrowns are the solution. They are strong and aesthetic. The dentists are happy to be able to offer such a beautiful restoration to their parents, parents are happy to have a healthy alternative to toxic metal crowns or weak strip crowns, and children are happy to be able to smile with confidence. In tandem with offering this solution, we offer Sprig University to teach the doctors how to successfully use them.
With USA being your main market, how do you look towards the rest of the world in terms of education, product delivery and serving your unique customer group?
Even though we started in the USA, our products have a universal benefit worldwide to the practice of dentistry and ultimately the children and their parents. We are currently expanding globally. It is important that we take the right approach with this expansion. The focus is on paediatric dentistry and the unique characteristics that the practice requires. We also look at each country closely so that we make sure we are adapting to any unique characteristics of the culture. Of course, product delivery is very important and along with that, serving our customers through the entire process of education, product implementation and beyond. As a medical device supplier, there are unique regulatory requirements for each country. We wish it was as easy as deciding to sell in a particular country. It’s not. The deciding is the easiest part, the real work comes after that decision.
Looking towards the middle east dentistry market, how is Sprig looking to interact and support the requirements of the paediatric dentistry needs in the region?
We have several long-time champions in the Middle East. We have had many doctors travel from the Middle East to take Sprig University. So, we knew there was potential. We then attended the February dental exhibition in Dubai, UAE last year and along with exhibiting, we sponsored a lecture on the use of zirconia crowns by Dr. Yasmin Kottait and held a hands-on training at our booth. The program we call Sprig University Mini. The response was overwhelming, not only from the doctors but the clinic administrators, distributors and representatives from just about every country in the Middle East.
We returned knowing we had more work to do to prepare for the demand in this market and fulfil the needs of the market. We have been preparing ever since. There are unique nuances with the Middle East and we feel we now have a better understanding on how to educate, provide customer care and ensure that we exceed their expectations on product quality and their experience with Sprig as a whole.
The Gulf is regarded amongst the youngest populations in the world, with Saudi Arabia having approximately 37% of their population under 14 years of age, how important is it for Sprig to enter the Gulf region?
Quality and innovative paediatric dentistry is driven by the parents of these children. In our experience, parents in the Gulf territories are even more progressive in their demands for healthier options for their children than other countries around the globe. With Sprig being the leader in providing these products to paediatric dentistry it is absolutely necessary for us to be in the Gulf region.
Having gone through the most unprecedented year in recent history; the global lockdown due to the Covid-19 pandemic, how has Sprig managed to survive 2020 so far?
These memories will be printed in my memory forever. I remember when dental offices were shutting down, state by state and country by country. As with everyone else, we did not know how severe this would be. We were patient but also proactive. As a medical device supplier, we were considered as an essential business. However, that did not give us any sort of immunity. We had to retrofit our offices, minimise the number of employees in the office at one time, and we did close for a brief period of time. We never actually stopped our shipping operations but the number of orders being processed and shipped drastically declined with the offices being closed.
Very early on, I gathered three of our managers to help strategise on how we could serve our doctors and community. How could we help? That was our main focus through the pandemic. We were here to serve. This service kept our team busy and our business moving in a forward direction. The goal was to get all the employees back to work as soon as possible. There were hardships but also great satisfaction in knowing we were helping our community and each other.
We miss travelling and seeing our customers in person. We have slowly started to add events and are very much looking forward to our visit to Dubai in March 2021. Technology has come a long way but there really is no better way to connect with people than in-person.
In 2021, IDS Cologne is scheduled to take place in September despite the ongoing global pandemic. During the show, majority of dental manufacturers are expected to introduce their novelties. What are your plans as Sprig and can we expect any product launches this year?
The pandemic gave us the time to step back and re-evaluate our goals. It also gave us the time to re-examine our current R&D projects. R&D is a constant activity at Sprig. The question was what the market wants and what does it need? We think we have solid answers to these questions, and we hope to be introducing several new products in the near future. We are also working very closely with our regulatory department, so we can introduce products globally and not just in the United States.
Thank you for the interview.
RAS AL-KHAIMAH, UAE: Although the association between dental anxiety and oral health-related quality of life is well established internationally, evidence ...
Dentsply Sirona is the world’s largest manufacturer of professional dental products and technologies. The company has been undergoing major restructuring,...
CAPP recently hosted an engaging pediatric hands-on course led by the esteemed Dr. LaRee Johnson from USA at the CAPP Training Centre in Dubai. The event, ...
Originally from Syria, Dr Iyad Estoiny obtained his master’s degree in fixed and removable prosthodontics in France before moving to Dubai in 1997. An...
The Straumann Group held Esthetic World 2022 in Dubai in January. In this interview, Dental Tribune MEA speaks to Wolfgang Becker, Executive Vice President,...
Dental Tribune MEA had the pleasure to speak with Ahmed Ghandour who is Sales Director of Neoss. He has a strong track record in the dental implant ...
BRUSSELS, Belgium: The European Federation of Periodontology (EFP) is inviting the global dental community to the EuroPerio10 congress in Copenhagen in ...
Dr Dobrina Mollova from Dental Tribune Middle East & Africa interviewed Dr Frank Thiel, Vice President Global Research and Development CAD/CAM and ...
Live webinar
Wed. 3 June 2026
9:00 pm UAE (Dubai)
Live webinar
Thu. 4 June 2026
10:00 pm UAE (Dubai)
Live webinar
Mon. 8 June 2026
8:00 pm UAE (Dubai)
Live webinar
Mon. 8 June 2026
9:00 pm UAE (Dubai)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
10:00 pm UAE (Dubai)
Live webinar
Wed. 10 June 2026
7:00 pm UAE (Dubai)
Live webinar
Wed. 10 June 2026
10:00 pm UAE (Dubai)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس



















































To post a reply please login or register